 
Calculate the number of magnesium atoms in 10.0g of magnesium. Let's try a different kind of calculation whichextends the ideas a little further. In all of the above examples we have determinedthe mass of a single atom, molecule or formula unit of asubstance. First the molecular mass of the compound must be determined for C: 12 x 12.0 u = 144 u for H: 22 x 1.01 u = 22.22 u for O: 11 x 16.0 u = 176 u Total = 342 u (molecular mass for one molecule of C 12H 22O 11) Now convert from units of 'u' to units of grams Calculate themass, in grams, of a molecule of sucrose, C 12H 22O 11. Now we can use a unitfactor to convert from u's to grams Now we can use a unitfactor to convert from u's to grams įinally, let's go ahead and calculate the massof a formula unit of magnesium chloride in grams The formula ofmagnesium chloride is MgCl 2 so its formula mass is24.305 u + 2 x 35.453 u which is 95.211 u. This is a very small number! Calculate the mass of a molecule of chlorine ingrams The formula of chlorine is Cl 2 so its molecularmass is 2 x 35.453 u which is 70.906 u. Now we can use a unit factor toconvert from u's to grams. From the periodictable this value is 24.305 u. First we canobtain the atomic mass of a magneium atom. Let's see how we can use this information tocalculate the mass of an atom of magnesium in grams. Now we have a way to determine the mass of a single atomof an element, a molecule of an element or compound and the massof a formula unit of an ionic compound. The 'u' is defined as one-twelfth of the mass of theisotope of 12C. Thatrelationship is given to us by the definition of the atomic massunit. We need arelationship between atomic mass units (u) and grams. But we still have a problem! The balances in thelaboratory measure mass in units of grams, not u's. Therefore the formula mass of MgCl 2 is 95.35u. We can obtain the mass of a formula unit of MgCl 2by simply summing the atomic masses for each of the atoms in theformula. The molecular mass forelemental chlorine, Cl 2, is 70.90 u, just two timesthe atomic mass. The formula forelemental chlorine is Cl 2. We usually shorten this and say theatomic mass of magnesium is 24.3 u. This is obtained by summing the product ofthe mass of each isotope of the element times the fractionalabundance of that isotope. 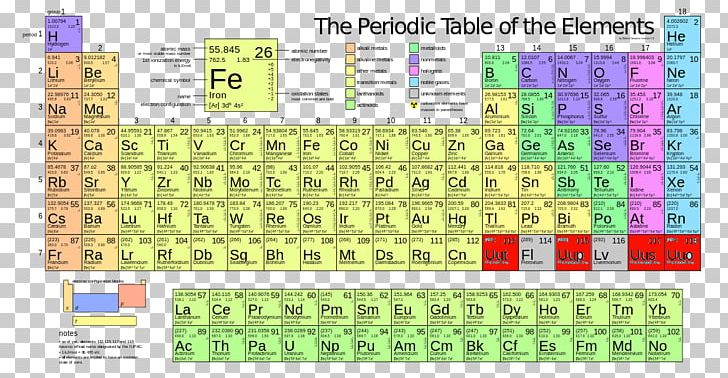 
The relative weighted average atomic mass formagnesium is 24.3 u. In the periodic table themass number for an element is given below the symbol. What is therelationship between the amount of substance and the number ofatoms or molecules? How are we able to perform quantitativechemical reactions? There is an answer! Lets return to the periodic table. In the chemistry laboratory wemust measure amounts of substances by mass. If we wanted to do this reaction toprepare some magnesium chloride it would be difficult to find asingle atom of magnesium and a single molecule of chlorine, evenfor the most experienced chemist. How do we read this equation? This equation reads 1 atom of magnesium combines with onemolecule of chlorine to form one formula unit of the ioniccompound magnesium chloride. We can balance the equation and we recognize it is an exampleof a formation reaction. Untitled Normal Page Consider the balanced equation 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
